OAT Coolant Inhibitors: Anti-Corrosion for Engines cooling
Leave a message
Are you interested in investing in automotive antifreeze production, but don't know where to start and how to produce it?
As a car owner, are you confused about how to choose from the various antifreezes on the market?
As an automotive antifreeze manufacturer, are you looking for low-cost, high-quality antifreeze corrosion inhibitor packages?
This article systematically explains the coolant/antifreeze issues you are concerned about.
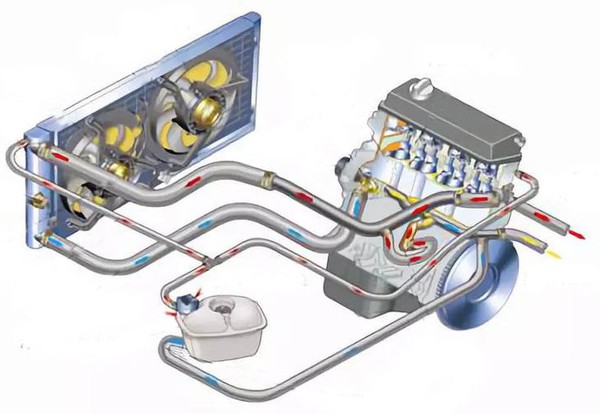
1. What is car antifreeze? Why do we need to add antifreeze to cars?
Antifreeze/coolant is a fluid containing special additives. It is mainly used in liquid-cooled engine cooling systems. It has excellent properties such as antifreeze in winter, anti-boiling in summer, anti-scaling, and anti-corrosion all year round.
5 core reasons why cars must use antifreeze/coolant.
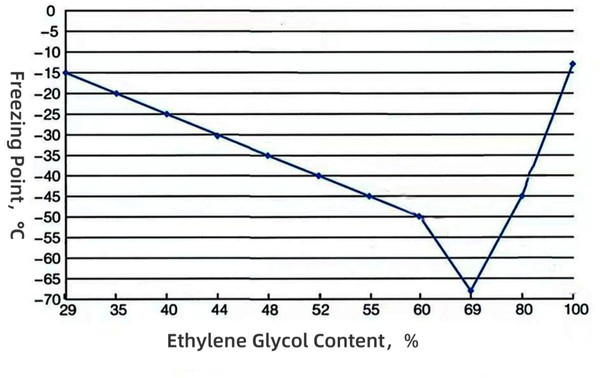
1. Antifreeze protection - 50% ethylene glycol aqueous solution has a freezing point of about- 37℃ (pure water freezes at 0℃), preventing the cylinder block/water tank from freezing and cracking in extreme cold weather.
2. High temperature boiling prevention - The boiling point of the same concentration solution is 107℃ (pure water boils at 100℃), avoiding high temperature "boiling", and adapting to the 105-120℃ operating temperature of modern engines (SAE J20 standard).
3. Corrosion inhibition - Corrosion inhibitors (Organic Acids Technology /OAT) form a passivation film on the metal surface to protect the corrosion rate of metal parts such as copper/aluminum.
4. Scale prevention - Use deionized water + additives to eliminate scale and maintain heat dissipation efficiency.
5. Water pump lubrication - Reduce mechanical wear and increase the life of aluminum pumps.
2. A brief history of automotive antifreeze
Simply, modern automotive ethylene glycol (MEG) -based antifreeze/coolant was mainly promoted for commercial use by Dow Chemical since the 1930s, from inorganic salts to OAT/HOAT, which is the foundation for modern antifreeze.
- Early stage (before 1920s): Water, methanol, or glycerol were used as cooling media in automotive coolants. Methanol is volatile, flammable, and toxic; glycerol has limited low-temperature performance (freezing point is about -17°C).
- Ethylene glycol MEG (1930s): In the 1930s, Dow Chemical in the United States took the lead in commercializing ethylene glycol (MEG) for automotive antifreeze. Its advantages are significant:
- Significantly lower freezing point (50% concentration freezing point ≈ minus 37°C)
- High boiling point (about 107°C).
- Large heat capacity and high cooling efficiency.
- Development of additive system (from 1940s): Early ethylene glycol was highly corrosive and required the addition of corrosion inhibitors. During World War II, the US military promoted the development of standardized formulas (such as adding borates, phosphates, silicates, etc.).
- After the 1970s, Organic Acid Technology (OAT) emerged to provide longer life protection
- Modern evolution: Continuous optimization of additive packages (low silicon/silicon-free, organic-inorganic hybrid technology HOAT), improved compatibility, environmental protection (propylene glycol-based products), and long-term effectiveness (5 years/250,000 kilometers).
- Antifreeze for electric vehicles: China's upcoming mandatory national standard GB29743.2 "Motor Vehicle Coolant Part 2: Electric Vehicle Coolant" clearly requires the use of low conductivity coolant (≤100 μS/cm), enhanced anti-corrosion performance, and high cleanliness level, and is scheduled to be promulgated in August 2025. The industry will complete the technology replacement in early 2026.
3. What are the main components of automotive antifreeze?
Core components: A water-based solution (SAE J1034 standard) with ethylene glycol/propylene glycol (accounting for 45%-60%) as the base liquid and additives such as corrosion inhibitors, defoamers, bitter, and dyes.
Its core function is to ensure efficient operation of the cooling system by regulating the freezing point/boiling point and corrosion protection of the solution.
Ethylene glycol: By mixing ethylene glycol (propylene glycol) and water in different proportions, an antifreeze suitable for ambient temperature can be obtained. Under normal circumstances, the freezing point of the selected antifreeze should generally be more than 10°C lower than the local minimum temperature to prepare for sudden weather changes.
Deionized water
Why does antifreeze have extremely high requirements for water quality?
Water is a good heat-absorbing carrier (large specific heat capacity), but ordinary tap water or groundwater must not be used! All international standards (such as ASTM D3306, etc.) stipulate that deionized water must be used to prepare antifreeze to completely remove these harmful impurities to ensure the performance and safety of the antifreeze.
These waters contain a large amount of minerals (calcium, magnesium, chlorine, sulfate, etc.). These impurities will cause scale, reduce heat dissipation efficiency, and, in severe cases, will block the cooling system pipes. At the same time, these impurities will accelerate corrosion, especially chlorine and sulfate, which will quickly corrode key metal parts of the engine, such as aluminum and iron.
The freezing point of antifreeze corresponding to different ratios of antifreeze (MEG) and water
Corrosion inhibitors
The engine and its cooling system are made of metal, including copper, iron, aluminum, steel, solder, and other materials. These metals will corrode and rust over time when in contact with water/ethylene glycol at high temperatures.
No corrosion inhibitor can protect all metals in the engine cooling system. Therefore, the difficulty of antifreeze research and development lies in finding a suitable composite formula containing multiple corrosion inhibitors, which can not only protect all metals in the system from corrosion, pitting, and cavitation, but also maintain stable performance in long-term use, while being cheap and environmentally friendly.
At present, there are three main types of composite formulas for antifreeze: inorganic, organic acid technology (OAT), and organic-inorganic composite ( Hybrid organic acid technology, HOAT).
The corrosion inhibitor package AC7040, based on organic acid technology designed by Chorus, has passed the SGS test and fully complies with the ASTM D3306 standard.
Antifreeze/Coolant Additive Package AC7040 (OAT)

The antifreeze/coolant on the market has various colors, mainly including blue, yellow, green, and red. These colors are produced by adding dyes. Dyes are only used to distinguish brands and eye-catching colors to provide leak alerts.
The quality and application range of antifreeze cannot be judged by color. Different colors do not represent any special functions.
5. Is antifreeze a high-tech product?
For traditional antifreeze, ethylene glycol and deionized water can be easily obtained from the local market, the market prices are very open, while the corrosion inhibitor formula is the core technology of the enterprise. If the antifreeze manufacturer does not have the ability to develop corrosion inhibitor compounds, it can also be obtained from professional corrosion inhibitor package suppliers. This type of compound should have the following characteristics:
Protection of multiple engine materials: The engine cooling system contains materials such as cast iron, aluminum alloy, copper and solder. The corrosion inhibitors in the formula should work synergistically to provide comprehensive protection for multiple metals. Generally, the formula that needs to pass the corresponding tests and market verification is the truly excellent formula.
Long-term stability: Modern organic acid technology (OAT) coolant forms a stable protective film, which can protect the engine cooling system for a long time, with a service life of up to 5 years or more than 200,000 kilometers. But the inorganic coolant should be replaced 1-2 years due to the inorganic salt being consumed
Environmental protection requirements: Traditional nitrites and phosphates are harmful to the environment, and developed countries in Europe and the United States have long banned the use of nitrites; although organic acid technology (OAT) has a higher cost, it is increasingly favored by global users due to its long-term and environmentally friendly nature.
6. What are the main standards for antifreeze?
Chinese standard
GB 29743.1-2022: Basic Standard for Coolant of Fuel Vehicles (Replacing GB 29743-2013)
NB/SH/T 0521-2010: Light Load Standard for Ethylene glycol type
JIS K2234: Japanese Standard (Stricter Requirements for Aluminum corrosion)
International standard
ASTM D3306: Standard for Passenger Vehicles (USA)
ASTM D4985: Heavy-duty Standard (including SCA Additives)
ISO 6525: Standard of the International Organization for Standardization
SAE J1034: Society of Automotive Engineers Standard
OEM standard
Volkswagen TL 774-C/G40
Mercedes-Benz 325.3/325.5
BMW GS 9400
7. What are the main testing standards for antifreeze?
Generally speaking, the main test items for antifreeze are divided into: basic physical and chemical properties, corrosion protection performance, chemical composition and stability, and other properties.
Basic physical and chemical properties
Appearance/color/smell: visual and olfactory detection (GB 29743.1 requires eye-catching colors)
Density (20℃): reflects concentration, standard ≥1.060g/cm³
Freezing point/boiling point: key protection index (e.g., -35℃ type requires ≤-35℃, boiling point ≥107.5℃)
pH value: 7.5-11.0 range, to prevent acid corrosion
Ash content: ≤2.5%, reflecting the content of inorganic salts
Corrosion protection performance
Glassware corrosion (88℃/336h): mass changes of 6 metal test pieces (such as copper ±10mg)
Simulated use corrosion (88℃/1064h): more stringent long-term corrosion test
Cast aluminum alloy heat transfer corrosion (135℃/168h): special test of high temperature and high pressure (±1.0mg/cm²)
Aluminum pump cavitation corrosion: ≥8 level (ASTM D2809 method)
Chemical stability
Chlorine content: ≤25mg/kg (chloride ions cause pitting)
Sulfate: ≤50mg/kg (SO₄²⁻ causes precipitation)
Reserve alkalinity: buffering acidic substances (reportable value)
Hard water stability: precipitation ≤0.5mL (anti-calcium and magnesium ion reaction)
Other properties
Foam tendency: volume ≤150mL and dissipates within 5 seconds
Effect on organic finishes: verify compatibility
Storage stability (60℃/336h): color and precipitation control






